EISAI TO PRESENT NEW DATA FROM LEQEMBI® (LECANEMAB-IRMB) PHASE 3 CLARITY AD STUDY AND OTHER ALZHEIMER’S DISEASE PIPELINE RESEARCH AT THE CLINICAL TRIALS ON ALZHEIMER’S DISEASE (CTAD) CONFERENCE
Update on LEQEMBI Investigational Subcutaneous Formulation
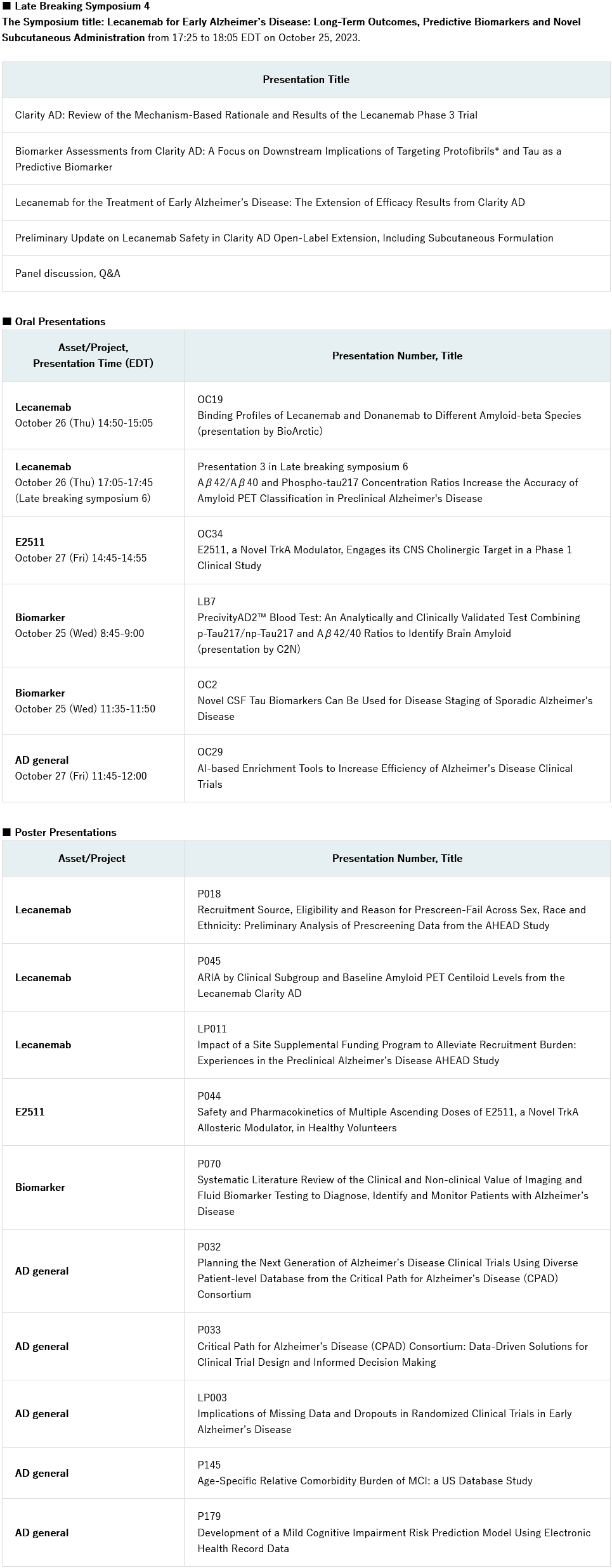
Eisai Co. Ltd (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”) announced today that the company will present new data from the phase 3 Clarity AD study for its Alzheimer’s disease (AD) treatment LEQEMBI® (lecanemab-irmb) 100 mg/mL injection for intravenous use and new data on the subcutaneous formulation in development at the 16th annual Clinical Trials on Alzheimer’s Disease (CTAD) conference. The conference will be held in Boston, Massachusetts, United States and virtually from October 24 to 27, 2023. In addition to the data presented on Eisai’s anti-amyloid beta (Aβ) protofibril* antibody LEQEMBI, phase 1 data for E2511, an investigational tropomyosin receptor Kinase A (TrkA) positive allosteric modulator (PAM), will be presented as well as other research from the company’s AD pipeline. At the conference, Eisai will present data and research in five oral and ten poster presentations. BioArctic will give an oral presentation on lecanemab.
Late-Breaking Symposium 4 – Lecanemab for early Alzheimer’s Disease: Long-Term Outcomes, Predictive Biomarkers, and Novel Subcutaneous Administration
- In a late-breaking symposium on October 25 from 17:25-18:05 EDT, Eisai will present the latest data from the Clarity AD optional tau PET longitudinal substudy. The presentation will include a post-hoc analysis of the low and intermediate + high-tau subgroups, with the low-tau subgroup representing early stages of disease studied specifically in the phase 3 core study, and the open-label extension study. An update on the investigational subcutaneous formulation, including interim safety and effect on amyloid in the brain measured by amyloid PET, will be provided.
- Distinguished faculty members Christopher van Dyck M.D., Keith Johnson M.D. and Reisa Sperling M.D. will discuss the findings in a panel led by Michael Irizarry, M.D., MPH, Eisai.
- A live webcast of this symposium can be viewed on the Eisai Co., Ltd. website.
“Alzheimer’s disease is a progressive and relentless condition that requires early diagnosis and continued treatment. LEQEMBI supports neuronal function in Alzheimer’s disease by clearing highly toxic protofibrils that can continue to cause neuronal injury and death well after plaques are cleared,” said Michael Irizarry, MD, MPH, Senior Vice President, Clinical Research, Neurology, Deputy Chief Clinical Officer, Clinical Evidence Generation, Eisai. “We look forward to sharing the new LEQEMBI low-tau subgroup data and subcutaneous data at CTAD 2023.”
Other major oral presentations include:
- Lecanemab: Binding profiles of lecanemab and donanemab to different amyloid-beta species (OC19, presentation by BioArctic).
- E2511, a novel TrkA modulator, engages its CNS cholinergic target in a phase 1 clinical study (OC34).
- Novel CSF tau biomarkers can be used for disease staging of sporadic Alzheimer’s disease (OC2).
The full list of presentations about Eisai assets and research follows.
This release discusses investigational uses of agents in development and is not intended to convey conclusions about efficacy or safety. There is no guarantee that such investigational agents will successfully complete clinical development or gain health authority approval.
MEDIA CONTACTS:
Eisai Co., Ltd.
Public Relations Department
TEL: +81 (0)3-3817-5120
Eisai Inc. (U.S.)
Libby Holman
+ 1-201-753-1945
Libby_Holman@Eisai.com
Eisai Europe, Ltd.
(UK, Europe, Australia, New Zealand and Russia)
EMEA Communications Department
+44 (0) 786 601 1272
EMEA-comms@eisai.net
