EISAI TO PRESENT PRECLINICAL AND CLINICAL RESEARCH ON ERIBULIN AT THE 2022 SAN ANTONIO BREAST CANCER SYMPOSIUM
Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”) announced today that new study results on its in-house discovered and developed anticancer agent eribulin mesylate (HALAVEN®, “eribulin”) will be presented during the 2022 San Antonio Breast Cancer Symposium (SABCS), which is taking place virtually and in-person in San Antonio, Texas from December 6-10.
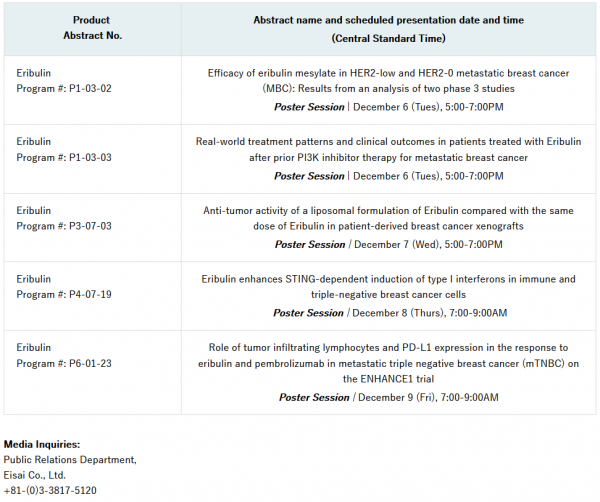
Eisai will present five eribulin-related abstracts, including a post hoc subgroup analysis from two pivotal Phase 3 studies (EMBRACE and Study 301), as well as:
– Real world use of eribulin following treatment with a P13K inhibitor, mostly in people with Hormone Receptor (HR)-positive/HER2-negative metastatic breast cancer.
– Preclinical data exploring a liposomal formulation of eribulin, in a Phase 1 expansion cohort for breast cancer, versus eribulin at the same dose, in patient-derived breast cancer xenografts.
“We continue to relentlessly pursue research that provides useful insights for people living with breast cancer,” said Dr. Takashi Owa, Chief Scientific Officer, Senior Vice President, Eisai Co., Ltd. “A big part of this commitment is the ongoing sharing of our preclinical and clinical data with eribulin.”
This release discusses investigational compounds and investigational uses for FDA-approved products. It is not intended to convey conclusions about efficacy and safety. There is no guarantee that any investigational compounds or investigational uses of FDA-approved products will successfully complete clinical development or gain FDA approval.
Eisai presentations at the 2022 SABCS are as follows: