Eisai and Merck Present First-Time Data From Two Studies Evaluating KEYTRUDA® (pembrolizumab) Plus LENVIMA® (lenvatinib) in Seven Different Tumor Types at ESMO Virtual Congress 2020
New Results Include Findings From the Phase 2 LEAP-004 Trial Showing an ORR of 21.4% in Patients With Unresectable or Advanced Melanoma Who Had Previously Progressed on an Anti-PD-1/PD-L1 Therapy
TOKYO and KENILWORTH, N.J. [September 23, 2020] – Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”) and Merck & Co., Inc., Kenilworth, N.J., U.S.A. (known as MSD outside the United States and Canada) announced new investigational data from two trials under the LEAP (LEnvatinib And Pembrolizumab) clinical program evaluating LENVIMA, the orally available multiple receptor tyrosine kinase inhibitor discovered by Eisai, plus KEYTRUDA, Merck’s anti-PD-1 therapy. In the Phase 2 LEAP-004 trial, the LENVIMA plus KEYTRUDA showed an objective response rate (ORR) of 21.4% (95% confidence interval (CI): 13.9-30.5) in patients with unresectable or advanced melanoma who had previously progressed on an anti-PD-1/PD-L1 therapy. In the Phase 2 LEAP-005 trial, LENVIMA plus KEYTRUDA demonstrated an ORR that ranged from 9.7-32.3% (95% CI: 2.0-51.4) in previously treated patients with triple-negative breast cancer (TNBC), ovarian cancer, gastric cancer, colorectal cancer (non-microsatellite instability-high [non-MSI-H]/mismatch repair proficient [pMMR]), glioblastoma multiforme (GBM) and biliary tract cancer (BTC). Results from LEAP-004 (Abstract #LBA44) and LEAP-005 (Abstract #LBA41) were accepted as late-breaking abstracts and are being presented in proffered paper presentations at the European Society for Medical Oncology (ESMO) Virtual Congress 2020.
“These new data from our LEAP clinical program show encouraging activity across several aggressive cancer types and expand our knowledge about the potential of KEYTRUDA plus LENVIMA to help a range of patients with these cancers,” said Dr. Scot Ebbinghaus, Vice President, Clinical Research, Merck Research Laboratories. “This is the first time that clinical data from two LEAP trials are being presented, reflecting important progress we are making to explore the potential of this combination for patients in need of new options, particularly those with advanced melanoma who have progressed on an anti-PD-1 or PD-L1 therapy.”
“We are encouraged by the growing body of research that we have seen to date, now in 13 different cancers, supporting the potential of the LENVIMA plus KEYTRUDA combination, which we’re currently evaluating in 19 clinical trials,” said Dr. Takashi Owa, Chief Medicine Creation and Chief Discovery Officer, Oncology Business Group at Eisai. “These data not only help advance our understanding of the regimen but also fuel our deep-seated determination to work to address the unmet needs of these patients.”
Lenvatinib (len) plus pembrolizumab (pembro) for advanced melanoma (MEL) that progressed on a PD-1 or PD-L1 inhibitor: initial results of LEAP-004 (Abstract #LBA44)
LEAP-004 (ClinicalTrials.gov, NCT03776136) is a Phase 2, single-arm, open-label trial evaluating LENVIMA in combination with KEYTRUDA in patients with unresectable or advanced melanoma who had progressed on an anti-PD-1/PD-L1 therapy within 12 weeks. Patients were treated with LENVIMA 20 mg orally once daily until unacceptable toxicity or disease progression in combination with KEYTRUDA 200 mg intravenously every three weeks for up to 35 cycles (approximately two years). The primary endpoint is ORR per Response Evaluation Criteria In Solid Tumors (RECIST) v1.1 as assessed by blinded independent central review (BICR). Secondary endpoints include progression-free survival (PFS) and duration of response (DOR) per RECIST v1.1 by BICR, overall survival (OS) and safety.
At data cutoff (June 10, 2020), a total of 103 patients were enrolled and treated. With a median duration of follow-up of 12 months (range: 8.7-15.6), LENVIMA plus KEYTRUDA demonstrated an overall ORR by BICR of 21.4% (n=22) (95% CI: 13.9-30.5), with a complete response rate of 1.9% (n=2) and a partial response rate of 19.4% (n=20). In the total study population, the median DOR was 6.3 months (range: 2.1+ to 11.1+), with 72.6% (95% CI: 46.2-87.6) of responses lasting for at least six months. Median PFS was 4.2 months (95% CI: 3.5-6.3), with 73.8% of patients experiencing disease progression or death, and the nine-month PFS rate was 26.2% (95% CI: 17.4-35.9). Median OS was 13.9 months (95% CI: 10.8-not reached [NR]), with death occurring in 44.7% of patients, and the nine-month OS rate was 65.4% (95% CI: 55.2-73.8).
The exploratory analysis showed that specifically, in the 29 patients whose disease progressed after an anti-PD-1/L1 therapy plus an anti-CTLA-4 therapy, the ORR by BICR was 31% (95% CI: 15.3-50.8), with a complete response rate of 3.4% (n=1) and a partial response rate of 27.6% (n=8). Disease control rate (DCR) by BICR in these patients was 62.1% (95% CI: 42.3-79.3). In the total study population, the DCR by BICR was 65% (95% CI: 55.0-74.2).
Treatment-related adverse events (TRAEs) led to discontinuation of LENVIMA and/or KEYTRUDA in 7.8% of patients. Grade 3-5 TRAEs occurred in 44.7% of patients (Grade 3: 39.8%; Grade 4: 3.9%; Grade 5: 1.0%), and serious TRAEs occurred in 18.4% of patients. The most common TRAEs of any grade occurring in at least 30% of the overall study population, were hypertension (56.3%), diarrhea (35.9%) nausea (34.0%), hypothyroidism (33.0%) and decreased appetite (31.1%).
LEAP-005: Phase 2 Study of Lenvatinib Plus Pembrolizumab in Patients (Pts) With Previously Treated Advanced Solid Tumors (Abstract #LBA41)
LEAP-005 (ClinicalTrials.gov, NCT03797326) is a Phase 2, single-arm, open-label trial evaluating LENVIMA in combination with KEYTRUDA in patients with select previously treated advanced solid tumors. The study cohorts are TNBC, ovarian cancer, gastric cancer, colorectal cancer (non-MSI-H/pMMR), GBM and BTC. Patients were treated with LENVIMA 20 mg orally once daily until unacceptable toxicity or disease progression in combination with KEYTRUDA 200 mg intravenously every three weeks for up to 35 cycles (approximately two years). The primary endpoints are ORR per RECIST v1.1 as assessed by BICR or Response Assessment in Neuro-Oncology (RANO) criteria (for GBM only) as assessed by BICR, and safety. Secondary endpoints include DCR per RECIST v1.1 by BICR or RANO (for GBM only) by BICR, DOR per RECIST v1.1 by BICR or RANO (for GBM only) by BICR, PFS per RECIST v1.1 by BICR or RANO (for GBM only) by BICR, and OS.
At data cutoff (April 10, 2020), a total of 187 patients were enrolled and treated. The confirmed ORR after a median duration of follow-up of 8.6 months (range: 1.9-13.1), for the six different tumor types, as well as additional efficacy and safety results, showed:
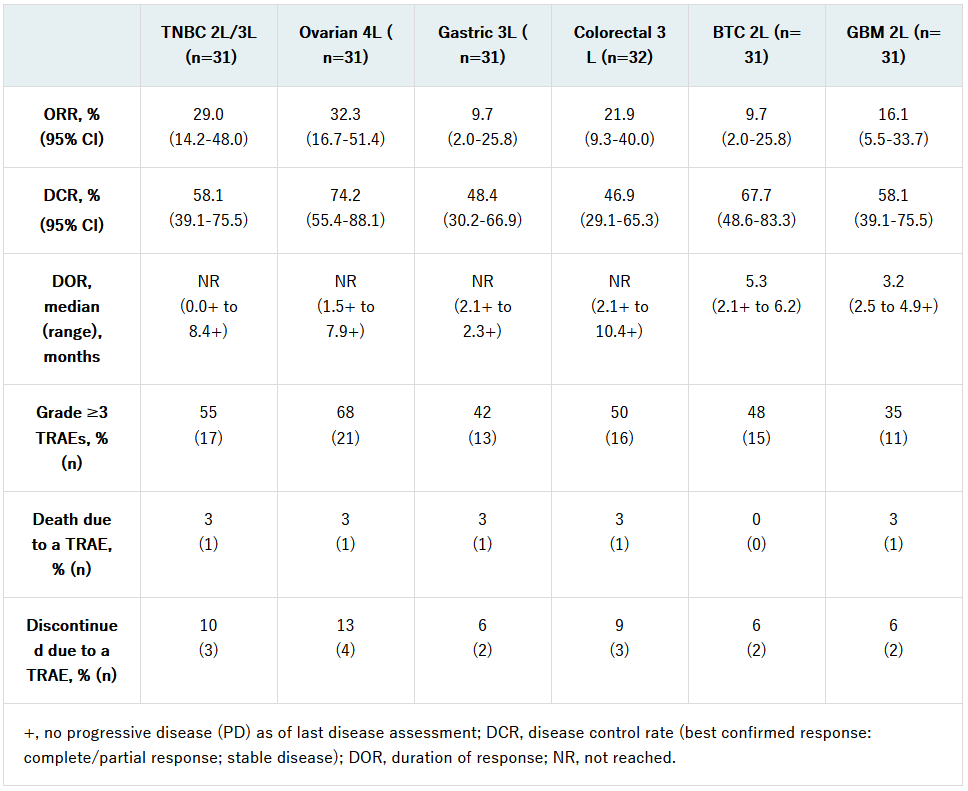
The most common TRAEs of any grade occurring in at least 20% of the overall study population were hypertension (39.0%), fatigue (29.4%), diarrhea (26.7%), decreased appetite (25.1%), hypothyroidism (27.8%) and nausea (21.9%). Based on these initial results, the trial will expand to enroll approximately 100 patients in each cohort.
About LENVIMA® (lenvatinib) Capsules
LENVIMA, discovered and developed by Eisai, is a kinase inhibitor that inhibits the kinase activities of vascular endothelial growth factor (VEGF) receptors VEGFR1 (FLT1), VEGFR2 (KDR), and VEGFR3 (FLT4). LENVIMA inhibits other kinases that have been implicated in pathogenic angiogenesis, tumor growth, and cancer progression in addition to their normal cellular functions, including fibroblast growth factor (FGF) receptors FGFR1-4, the platelet derived growth factor receptor alpha (PDGFRα), KIT, and RET. In syngeneic mouse tumor models, lenvatinib decreased tumor-associated macrophages, increased activated cytotoxic T cells, and demonstrated greater antitumor activity in combination with an anti-PD-1 monoclonal antibody compared to either treatment alone. Currently, LENVIMA has been approved for monotherapy as a treatment for thyroid cancer in over 65 countries including Japan, the United States, in Europe, and in Asia, and for unresectable hepatocellular carcinoma in over 65 countries including Japan, the United States, in Europe, China and in Asia. Additionally, it is also approved in combination with everolimus as a treatment for renal cell carcinoma following prior antiangiogenic therapy in over 55 countries, including the United States, in Europe (where it was launched under the brand name Kisplyx® for renal cell carcinoma) and in Asia. In addition, it is approved in combination with KEYTRUDA as a treatment for patients with advanced endometrial cancer that is not microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), who have disease progression following prior systemic therapy and are not candidates for curative surgery or radiation in countries including the United States, Australia, and Canada. Continued approval for this indication is contingent upon verification and description of clinical benefit in the confirmatory trials.
About KEYTRUDA® (pembrolizumab) Injection
KEYTRUDA is an anti-PD-1 therapy that works by increasing the ability of the body’s immune system to help detect and fight tumor cells. KEYTRUDA is a humanized monoclonal antibody that blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2, thereby activating T lymphocytes which may affect both tumor cells and healthy cells.
Merck & Co., Inc., Kenilworth, N.J., U.S.A. has the industry’s largest immuno-oncology clinical research program. There are currently more than 1,200 trials studying KEYTRUDA across a wide variety of cancers and treatment settings. The KEYTRUDA clinical program seeks to understand the role of KEYTRUDA across cancers and the factors that may predict a patient’s likelihood of benefitting from treatment with KEYTRUDA, including exploring several different biomarkers.
About the Eisai and Merck & Co., Inc., Kenilworth, N.J., U.S.A. Strategic Collaboration
In March 2018, Eisai and Merck, known as MSD outside the United States and Canada, through an affiliate, entered into a strategic collaboration for the worldwide co-development and co-commercialization of LENVIMA. Under the agreement, the companies will jointly develop, manufacture and commercialize LENVIMA, both as monotherapy and in combination with Merck’s anti-PD-1 therapy KEYTRUDA.
In addition to ongoing clinical studies evaluating the KEYTRUDA plus LENVIMA combination across several different tumor types, the companies have jointly initiated new clinical studies through the LEAP (LEnvatinib And Pembrolizumab) clinical program and are evaluating the combination in 13 different tumor types (endometrial carcinoma, hepatocellular carcinoma, melanoma, non-small cell lung cancer, renal cell carcinoma, squamous cell carcinoma of the head and neck, urothelial cancer, biliary tract cancer, colorectal cancer, gastric cancer, glioblastoma, ovarian cancer and triple-negative breast cancer) across 19 clinical trials.
Eisai’s Focus on Cancer
Eisai focuses on the development of anticancer drugs, targeting the tumor microenvironment (with experience and knowledge from Halaven® (Eribulin mesylate) and Lenvima) and the driver gene mutation and aberrant splicing (leveraging RNA Splicing Platform) as areas (Ricchi) where real patient needs are still unmet, and where Eisai can become a frontrunner in oncology. Eisai will discover innovative new drugs with new targets and mechanisms of action from these Ricchi, with the aim of contributing to the cure of cancers.
About Eisai
Eisai is a leading global research and development-based pharmaceutical company headquartered in Japan, with approximately 10,000 employees worldwide. We define our corporate mission as “giving first thought to patients and their families and to increasing the benefits health care provides,” which we call our human health care(hhc) philosophy. We strive to realize our hhc philosophy by delivering innovative products in therapeutic areas with high unmet medical needs, including Oncology and Neurology. In the spirit of hhc, we take that commitment even further by applying our scientific expertise, clinical capabilities and patient insights to discover and develop innovative solutions that help address society’s toughest unmet needs, including neglected tropical diseases and the Sustainable Development Goals.
For more information about Eisai, please visit www.eisai.com (for global), us.eisai.com(for U.S.) or www.eisai.eu (for Europe, Middle East, Africa), and connect with us on Twitter (U.S. and global) and LinkedIn (for U.S.).
Merck & Co., Inc., Kenilworth, N.J., U.S.A.’s Focus on Cancer
Our goal is to translate breakthrough science into innovative oncology medicines to help people with cancer worldwide. At Merck & Co., Inc., Kenilworth, N.J., U.S.A., the potential to bring new hope to people with cancer drives our purpose and supporting accessibility to our cancer medicines is our commitment. As part of our focus on cancer, Merck & Co., Inc., Kenilworth, N.J., U.S.A. is committed to exploring the potential of immuno-oncology with one of the largest development programs in the industry across more than 30 tumor types. We also continue to strengthen our portfolio through strategic acquisitions and are prioritizing the development of several promising oncology candidates with the potential to improve the treatment of advanced cancers. For more information about our oncology clinical trials, visit www.merck.com/clinicaltrials.
About Merck & Co., Inc., Kenilworth, N.J., U.S.A.
For more than 125 years, Merck & Co., Inc., Kenilworth, N.J., U.S.A., known as MSD outside of the United States and Canada, has been inventing for life, bringing forward medicines and vaccines for many of the world’s most challenging diseases in pursuit of our mission to save and improve lives. We demonstrate our commitment to patients and population health by increasing access to health care through far-reaching policies, programs and partnerships. Today, Merck & Co., Inc., Kenilworth, N.J., U.S.A. continues to be at the forefront of research to prevent and treat diseases that threaten people and animals – including cancer, infectious diseases such as HIV and Ebola, and emerging animal diseases – as we aspire to be the premier research-intensive biopharmaceutical company in the world. For more information, visit www.merck.comand connect with us on Twitter, Facebook, Instagram, YouTube and LinkedIn.
Forward-Looking Statement of Merck & Co., Inc., Kenilworth, N.J., U.S.A.
This news release of Merck & Co., Inc., Kenilworth, N.J., U.S.A. (the “company”) includes “forward-looking statements” within the meaning of the safe harbor provisions of the U.S. Private Securities Litigation Reform Act of 1995. These statements are based upon the current beliefs and expectations of the company’s management and are subject to significant risks and uncertainties. There can be no guarantees with respect to pipeline products that the products will receive the necessary regulatory approvals or that they will prove to be commercially successful. If underlying assumptions prove inaccurate or risks or uncertainties materialize, actual results may differ materially from those set forth in the forward-looking statements.
Risks and uncertainties include but are not limited to, general industry conditions and competition; general economic factors, including interest rate and currency exchange rate fluctuations; the impact of the recent global outbreak of novel coronavirus disease (COVID-19); the impact of pharmaceutical industry regulation and health care legislation in the United States and internationally; global trends toward health care cost containment; technological advances, new products and patents attained by competitors; challenges inherent in new product development, including obtaining regulatory approval; the company’s ability to accurately predict future market conditions; manufacturing difficulties or delays; financial instability of international economies and sovereign risk; dependence on the effectiveness of the company’s patents and other protections for innovative products; and the exposure to litigation, including patent litigation, and/or regulatory actions.
The company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events or otherwise. Additional factors that could cause results to differ materially from those described in the forward-looking statements can be found in the company’s 2019 Annual Report on Form 10-K and the company’s other filings with the Securities and Exchange Commission (SEC) available at the SEC’s Internet site (www.sec.gov).
