EISAI PRESENTS RESULTS OF PHASE III TRIAL OF LENVIMA® (LENVATINIB) IN UNRESECTABLE HEPATOCELLULAR CARCINOMA IN ORAL SESSION AT 20TH CSCO ANNUAL MEETING
RESULTS OF SUBPOPULATION ANALYSIS OF PATIENTS FROM GREATER CHINESE REGION (MAINLAND CHINA, HKSA, AND CHINESE TAIWAN)
Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”) has announced that the results of a subpopulation analysis of patients from the Greater Chinese Region (mainland China, HKSA, and Chinese Taiwan) in a Phase III trial (REFLECT / Study 304) of its in-house discovered and developed anticancer agent lenvatinib mesylate (product names: Lenvima® / Kisplyx®, “lenvatinib”) versus sorafenib as a first-line treatment for unresectable hepatocellular carcinoma (HCC) were orally presented for the first time during the 20th Annual Meeting of the Chinese Society of Clinical Oncology (CSCO), which took place in Xiamen (Amoy) in the Fujian Province of China. Over half of the world‘s HCC patients come from the Greater Chinese Region.
In the subpopulation analysis, lenvatinib demonstrated efficacy based on extension of Overall Survival (OS) compared to sorafenib (nominal P = 0.026), with improvements also observed in Progression Free Survival (PFS), Time to Progression (TTP) and Objective Response Rate (ORR) (see table below). Approximately 80% of patients in the subpopulation were suffering from HCC resulting from chronic hepatitis B virus (HBV). For these patients, median OS in the lenvatinib group (123 patients) was 14.9 months, compared to 9.9 months in the sorafenib group (119 patients) (Hazard Ratio [HR] 0.72, 95% Confidence Interval [CI] = 0.53-0.97). These findings are consistent with the overall results of the Greater Chinese Region subpopulation.
HBV is considered to be a negative predictor of tumor response to existing drug therapies. However, this data supports the effect of lenvatinib in patients with HCC resulting from HBV. Since there are many patients suffering from HCC resulting from HBV in the Greater Chinese Region, lenvatinib is expected to be a new treatment option for HCC patients in this area.
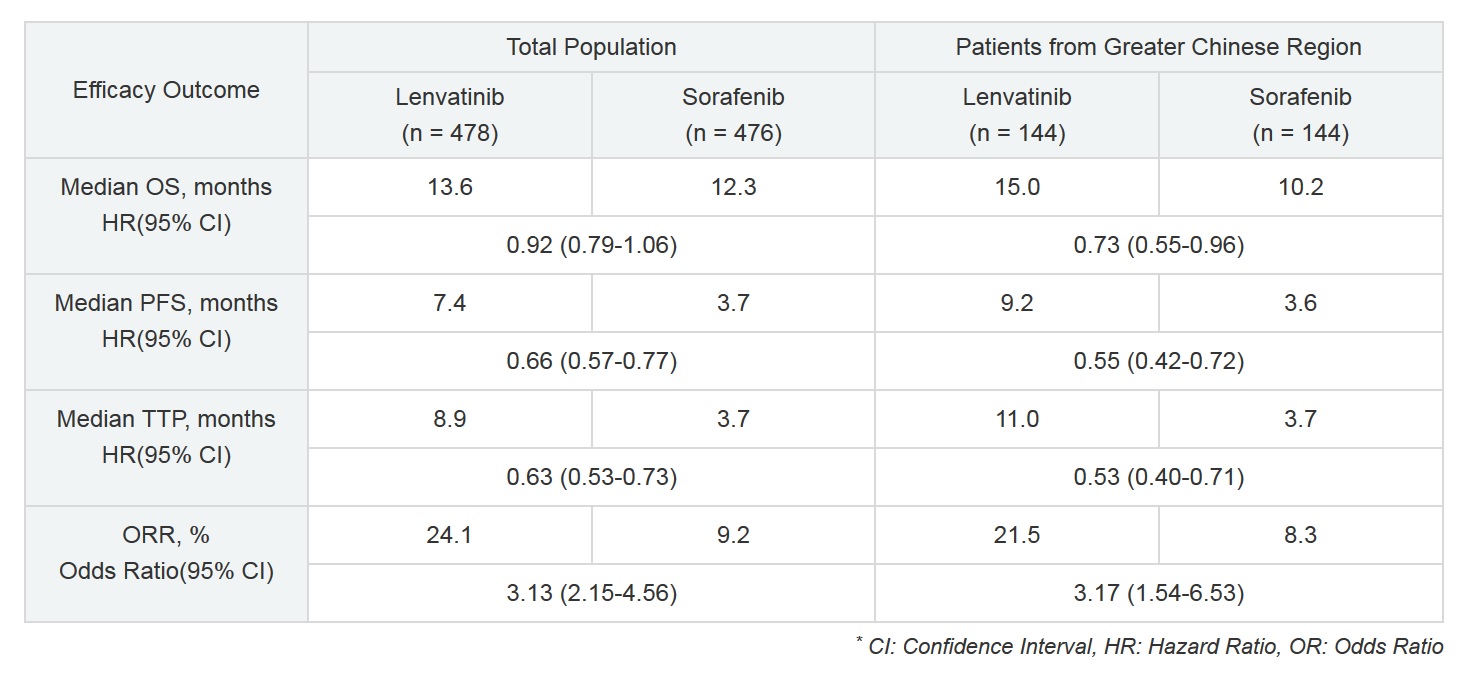
Additionally, lenvatinib’s safety profile for the Greater Chinese Region subpopulation was consistent with previous studies.
Liver cancer is the second leading cause of cancer related deaths and is estimated to be responsible for approximately 750,000 deaths per year globally. Additionally, approximately 780,000 cases are newly diagnosed each year, about 80% of which occur in Asian regions. Specifically, in China, there are approximately 395,000 new cases and 380,000 deaths per year, accounting for approximately 50% of cases worldwide. HCC accounts for 85% to 90% of primary liver cancer cases. Treatment options for unresectable HCC are limited and the prognosis is very poor, making this an area of high unmet medical need.
Following submissions in Japan (June 2017), the United States and Europe (July 2017), Eisai will submit a regulatory application for lenvatinib in HCC in China within the latter half of fiscal 2017. Eisai remains committed to generating scientific evidence aimed at maximizing the value of lenvatinib as it seeks to contribute further to addressing the diverse needs of, and increasing the benefits provided to, patients with cancer, their families, and healthcare providers.
